01 Introduction to Chemistry
02 The Structure of Atoms
2.1 The Basic Concept of Matter
2 Lessons
03 Concept of Mole, Formulae and Equations
3.2 Concept of Mole
3 Lessons
3.3 Chemical Formulae
2 Lessons
04 Periodic Table of Elements
4.5 Group 17 Elements
2 Lessons
5.3 Covalent Bonds
1 Lesson
5.2.1 Formation of Ion
Formation of Ion
- An ion is an atom or group of atoms carrying a positive or negative charge. Example Ca2+, O2-, SO42- etc.
- If a particle has equal numbers of protons (+) and electrons (-), the particle charge is zero, and the particle is said to be neutral.
- In a chemical reaction, electron(s) can be transferred from atom to another atom.
- If electron(s) is removed from an atom, the number of protons will be more than the number of electrons. In this case, the atom will have excess positive charge and hence form a positive ion (cation).

(An atom losses electron to form positive ion) - If an atom gains electrons, there is an excess negative charge in the atom, so a negative ion is formed.
- In other words,
- The atom losing electrons forms a positive ion (cation) and is usually a metal.
- The atom gaining electrons forms a negative ion (anion) and is usually a non-metallic element.
Recommended Videos
What are Ions | The Fuse School
What is an ion? What role does it have to play in the structure of atoms? Find out in this Chemistry Journey video by The Fuse School.
Formation of Positive Ion
- Usually, the atom of metals consists of 1, 2 or 3 valence electrons.
- In order to achieve octet electrons arrangement, the atoms will release the valence electrons.
- After releasing the valence electrons, the protons in the nucleus will outnumber the electrons. As a result, positive ions formed.
Example: Formation of Ion with +1 Charge
 |
| (A sodium atom release one electron to form a sodium ion with +1 charge) |
Example: Formation of Ion with +2 Charge
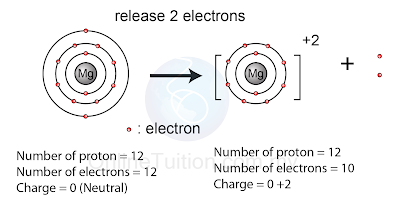 |
| (A magnesium atom release one electron to form a sodium ion with +2 charge) |
Example: Formation of Ion with +3 Charge
 |
| (An aluminium atom release one electron to form a sodium ion with +3 charge) |
Formation of Negative Ion
- Negative ions are formed among atoms of non-metal elements except group-18 elements.
- Non-metal elements usually located in group 15, 16 and 17 in the periodic table and have 5, 6 or 7 valence electrons.
- To achieve octet arrangement of electrons, their atoms will receive 3, 2 or 1 electron.
- As a result, the electrons will outnumber the protons and hence negative ions formed.
Formation of Negative Ions of -1 Charge
 |
| (A fluorine atom receives one electron to form a fluoride ion with -1 charge) |
Formation of Negative Ions of -2 Charge
 |
| (An oxygen atom receives two electrons to form an oxide ion with -2 charge) |
Difference Between an Atom and an Ion That Have Same Electrons Arrangement
Difference Between Fluoride Ion and Neon Atom
- After forming an ion, the electron arrangement of the ion is similar to the noble gases.
- For example, the electron arrangement of fluoride ion is 2.8 which is similar to a Neon atom, 2.8.
- Fluoride ion and the neon atom have similar electron arrangement.
- Fluoride ion carries charge whereas neon atom is neutral.
 |
| (The electrons arrangement of a fluoride ion is the same as a neon atom. However, they are so much different chemically) |
