01 Introduction to Chemistry
02 The Structure of Atoms
2.1 The Basic Concept of Matter
2 Lessons
03 Concept of Mole, Formulae and Equations
3.2 Concept of Mole
3 Lessons
3.3 Chemical Formulae
2 Lessons
04 Periodic Table of Elements
4.5 Group 17 Elements
2 Lessons
5.3 Covalent Bonds
1 Lesson
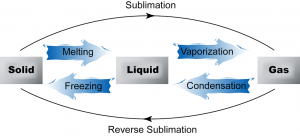
2.1.2 The Three States of Matter
3 States of Matter
Matter exists in 3 states of matter, namely, solid state, liquid state and gaseous state.
Characteristics of Matter in Solid, Liquid and Gaseous State
Arrangement of Particles
| Solid |
 |
| Particles are arranged in an orderly manner and close to one another. |
| Liquid |
 |
| Particles are not arranged in order. The space between particles is moderately large. |
| Gas |
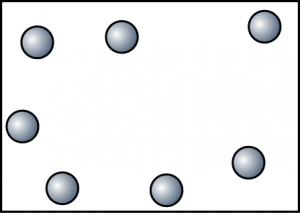 |
| The particles are very far apart and randomly arranged. |
Movement of Particles
| Solid | Particles vibrate at fixed positions. |
| Liquid | Particles move randomly and slowly and sometimes will collide against each other. |
| Gas | The particles move randomly in all directions at great speed. |
The force of Attraction Between Particles
| Solid | very strong |
| Liquid | Strong but weaker than in the solid state. |
| Gas | very weak |
Ability to be compressed
| Solid | Very difficult to be compressed because the particles are packed closely. |
| Liquid | Not easily compressed because the particles are packed quite closely. |
| Gas | Easily compressed because the particles are very far apart. |
Heat Energy content
| Solid | Lowest Energy Content |
| Liquid | Moderate energy content. |
| Gas | Highest energy content |
Volume and Shape
| Volume | Shape | |
| Solid | Fixed | Fixed |
| Liquid | Fixed | Follows the container |
| Gas | Follows the container | Fills the whole container |
Inter-conversion between the States of Matter
Change in Heat and Kinetic Energy of Particles
- The change in temperature will influences the kinetic energy or the speed of the motion of the particles.
- When a substance is heated, the kinetic energy of the particles in the substance increases. This causes the particles to move or vibrate faster.
- Likewise, when a substance is cooled, the kinetic energy of the particles in the substance decreases. This causes the particles to move or vibrate slower.
- The kinetic energy of the particles in a substance is directly proportional to the temperature of the substance.
Inter-conversion between States of Matter
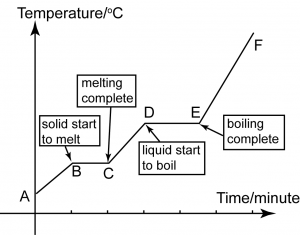
Heating Curve
A
A-B
B
B-C
C
C-D
D
D-E
E
E-F
A
- Naphthalene is in solid state at any temperature below its melting point.
- The particles are very closely packed together in an orderly manner.
- The forces between the particles are very strong. The particles can only vibrate at a fixed position.
A-B
- As the naphthalene is heated, heat energy is converted to kinetic energy.
- Kinetic energy increases and the molecules vibrate faster about their fixed positions and the temperature increases.
B
- Naphthalene is still in solid state.
- Naphthalene molecules have received enough energy to overcome the forces of attraction between them.
- Some of the particles that gain enough energy begin to move freely.
- Naphthalene starts to melt and changes into a liquid.
B-C
- Naphthalene exists in both solid and liquid states.
- The temperature remains constant because the heat that supplied to naphthalene is used to overcome the forces of attraction that hold the particles together.
- The constant temperature is called the melting point.
- The heat energy that absorbed to overcome the intermolecular forces is named as the latent heat of fusion.
C
- All the naphthalene has completely melted.
- Solid naphthalene has turned into liquid.
C-D
- Naphthalene is in liquid state.
- As the liquid naphthalene is heated, the molecules gain more heat energy and the temperature continues to increase.
- The particles move faster and faster because their kinetic energy is increasing.
D
- Naphthalene still exists in liquid state.
- Naphthalene molecules have received enough energy to overcome the forces of attraction between the particles in the liquid.
- Some of the naphthalene molecules start to move freely and liquid naphthalene begin to change into gas.
D-E
- Naphthalene exists in both liquid and gaseous states.
- The temperature remains unchanged.
- The is because the heat energy absorbed is used to overcome the intermolecular forces between the particles of the liquid rather than increase the temperature of the liquid.
- This constant temperature is the boiling point.
E
- All the naphthalene has turn into gas.
E-F
- The gas particles continue to absorb more energy and move faster.
- The temperature increases as heating continues.
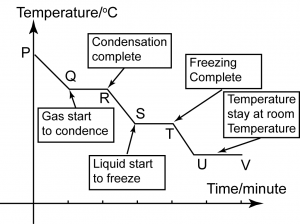
Cooling Curve
P
P-Q
Q
Q-R
R
R-S
S
S-T
T
T-U
U
P
- The substance exists in gaseous state.
- The particles have very high energy and are moving randomly.
- The intermolecular forces between the particles are very weak and can be ignored.
P-Q
- The substance is in gaseous state.
- The particles lose kinetic energy during cooling, the particles getting closer to each other and the temperature drops.
Q
- The substance still exists as a gas.
- As the molecules are close enough, stronger forces of attraction result in forming of intermolecular bonds.
- The gas begins to condense and become liquid.
Q-R
- The process of condensation going on.
- Stronger bonds form as gas changes into liquid.
- The substance exists in both gaseous and liquid states.
- The temperature remains unchanged.
- This is because the energy produced during the formation of bonds is equal to the heat energy released to the surroundings during cooling.
- This constant temperature is the boiling point.
- The heat energy that releases during this condensation process is called the latent heat of vaporization.
R
- The substance exists only in liquid state as all the gas particles have condensed into liquid.
R-S
- The substance exists as a liquid.
- As the temperature falls, the naphthalene molecules lose heat energy. Their movement shows down and they move closer to each other.
S
- The substance still in liquid state.
- The particles have very little energy and begin to move closer towards one another as it starts to freeze into solid.
S-T
- The liquid is changing into solid form.
- Molecules rearrange to form the molecular arrangement of a solid.
- The substance exists as both liquid and solid.
- The temperature remains constant until all the liquid changes to solid.
- This is because the energy released is the same as the energy lost to the surroundings during cooling.
- This constant temperature is the freezing point.
- The heat energy that releases during this freezing process is called the latent heat of fusion.
T
- All the liquid freezes into solid. The particles are now closely packed in an orderly manner.
T-U
- Once all the liquid has become solid, the temperature falls once again until it reaches room temperature. The substance is in the solid state here.
U
- The substance reaches room temperature and remain at this temperature as long as the room temperature remain the same.
Melting Point, Boiling Point and State of Matter

- The physical state of a substance at a certain temperature and pressure depends on the values of its melting and boiling points.
- A substance is in solid state if it exists at a temperature below its melting point.
- A substance is in liquid state if it exists at a temperature above its melting point but below its boiling point.
- A substance is in gaseous state if it exists at a temperature above its boiling point.