6.4.1 Chemical Properties of Acids
Acid + Reactive Metals
- Acids react with metals that is more electropositive than hydrogen in the electrochemical series to produce salt and hydrogen gas.
- Acids do not react with copper and silver.
- This is actually a displacement, where the metals that are placed above hydrogen in Electrochemical Series displace hydrogen from acid.
Example:
Example of Experiment
Procedure
- About 5 cm³ of dilute sulphuric acid is poured into a test tube.
- One spatula of zinc powder is added into the acid.
- A burning wooden splinter is placed at the mouth of the test tube.
- The observations are recorded.
Observation
Colourless gas is released. A “pop” sound produced when the wooden splinter is placed at the mouth of the test tube.
Inference
Hydrogen gas is released
Chemical Equation
H2SO4 + Zn → ZnSO4 + H2
Acid + Carbonate
Acids react with metal carbonates produces salt, water and carbon dioxide
Example:
Example of Experiment
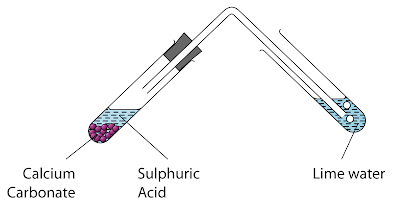
Procedure
- About 5 cm³ of dilute sulphuric acid is poured into a test tube.
- One spatula of calcium carbonate powder is added into the test tube.
- The gas released is passed through lime water as shown in the diagram above.
- The observations are recorded
Observation
Colourless gas is released. The gas turn lime water chalky.
Inference
Sulphuric acid reacts with calcium carbonate.
The gas released is carbon dioxide.
Chemical Equation:
H2SO4 +CaCO3 → CaSO4 +CO2 + H2O
Acid + Base Oxide
Acids react with bases produces salt and water
Example:
Hydrochloric acid + Magnesium Oxide
Example of Experiment
Procedure
- One spatula of copper(II) oxide powders is added to 5cm³ of hydrochloric acid in a test tube.
- The mixture is heated slowly and stirred with a glass rod.
- The observations are recorded.
Observation
- The black solid dissolves.
- The colourless solution turns blue.
Inference
- The copper(II) oxide powder (the black powder) has reacted with the hydrochloric acid.
- The blue colour solution contain copper(II) ions.
Chemical Equation
2HCl + CuO → CuCl2 + H2O
Acid + Alkali
- Acids react with alkali produces salt and water only.
- This is called a neutralisation reaction.
Example:
Ethanoic Acid + Sodium Hydroxide
CH3COOH + NaOH → CH3COONa + H2O
Hydrochloric Acid + Ammonia Solution
HCl + NH3 → NH4Cl