05 Chemical Bonds
06 Acids, Bases and Salts
6.5 Concentration of Aqueous Solution
2 Lessons
6.9 Preparation of Salts
4 Lessons
6.11 Qualitative Analysis
3 Lessons
7.1 Determining The Rate of Reaction
2 Lessons
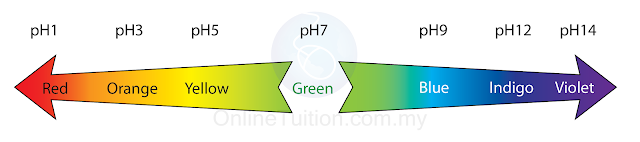
6.2.1 Meaning of pH and Determination of pH
The pH Scale
- pH scale is a measure of acidity and alkalinity. Practically, it is numbered between 0 and 14.
- Neutral substance has pH of 7.
- A solution of pH less than 7 is acidic The lower the pH, the more acidic the solution or the higher the concentration of H+ ions in the solution.
- A solution of pH above 7 is alkaline. The higher the pH, the more alkaline the solution or higher concentration of OH- ions presence in the solution.
- The pH of a solution can be measured by using the Universal indicator, pH paper or with a pH-meter.
Acids / Alkali Indicator
- An indicator is a substance or mixture of substances that when added to the solution gives different colours depending on the pH of the solution. Table below shows the colour of indicators in neutral, acidic and alkaline solution.Indicatorcolour in acid pH<7colour in neutral pH=7colour in alkali pH >7litmusred‘purple’bluephenolphthaleincolourlesscolourlesspinkmethyl orangepinky redorange about pH 6yellowmethyl redredorangeyellowbromothymol blueyellowgreenblue
- The best indicator of all is the universal indicator.
- It can be used to show not only whether a substance is acidic or alkaline, but also whether it is a strong acid or a weak acid.
 |
| (Colour of indicator at different pH value) |
| (Litmus paper. The colour of litmus is blue in alkali and red in acid) |
 |
| (Colour of phenolphthalein in alkali) |
| (Methyl Orange: The colour of mthyl orange in acidic and alkaline solution. Left: Acidic; Right: Alkaline. This image is shared by Rubashkyn in wikipedia) |
 |
| (Methyl Red: left: acidic, middle: neutral, right: alkaline. This image is shared by LHcheM in Wikipedia) |
 |
| (Bromothymol Blue pH indicator dye in an acidic, neutral, and alkaline solution (left to right).) |
 |
| (A roll of universal indicator paper. This image is shared by Bordercolliez in wikipedia) |
