02 Electrolysis
Exercise Summary
0 of 6 questions completed
Questions:
- 1
- 2
- 3
- 4
- 5
- 6
Information
You have already completed the exercise before. Hence you can not start it again.
Exercise is loading…
You must sign in or sign up to start the exercise.
You must first complete the following:
Results
Results
0 of 6 questions answered correctly
Time has elapsed
You have reached 0 of 0 point(s), (0)
Earned Point(s): 0 of 0, (0)
0 Essay(s) Pending (Possible Point(s): 0)
| Average score |
|
| Your score |
|
Categories
- Not categorized 0%
| Pos. | Name | Entered on | Points | Result |
|---|---|---|---|---|
| Table is loading | ||||
| No data available | ||||
- 1
- 2
- 3
- 4
- 5
- 6
- Answered
- Review
-
Question 1 of 6
1. Question
1 point(s)CHEM0602010
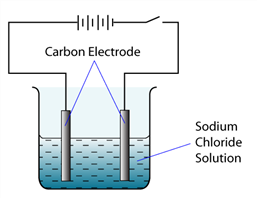
The diagram shows the set-up of the apparatus for an electrolytic cell. Which of the following ions are attracted to anode and cathode?
CorrectIncorrect -
Question 2 of 6
2. Question
1 point(s)Which of the following occur when molten silver bromide is electrolysed?
CorrectIncorrect -
Question 3 of 6
3. Question
1 point(s)In the electrolysis of dilute potassium sulphate solution by using carbon electrodes, what is the product formed around anode and cathode?
CorrectIncorrect -
Question 4 of 6
4. Question
1 point(s)Which of the following is formed at the anode during the electrolysis of dilute sodium chloride solution?
CorrectIncorrect -
Question 5 of 6
5. Question
1 point(s)A concentrated potassium chloride solution is electrolysed by using platinum electrodes. Which of the following half equation represent the reaction at anode and cathode?
CorrectIncorrect -
Question 6 of 6
6. Question
1 point(s)
The figure shows the apparatus set up for the electrolysis of potassium iodide solution and molten potassium iodide with carbon electrodes. What is the product formed in the anode of Cell 1 and Cell 2?
CorrectIncorrect